Keep Your Eyes on Ophthalmology
A Closer Look into Infectious Diseases, Devices and Disinfectants
Keywords
Endogenous – Growing or originating within an organism
Epibulbar – Situated on the eye
Exogenous – Growing or originating outside an organism
Ophthalmology – A branch of medicine and surgery which deals with the diagnosis and treatment of eye disorders
Pathogen – A microorganism that can cause disease
Parasitic/Protozoan Cysts – The more resistant form that parasites/protozoa take when under stress or in an unfavourable environment
The Spaulding Classification System – Developed by Dr. Earle H. Spaulding in 1968, this classification scheme defined how an item (e.g. medical device) should be disinfected based on where it is used in or on the body.
Abstract
Within the ophthalmological and optometry sector, eye care professionals are obligated to ensure the highest level of infection control is adhered to for the safety of patients and staff.
Microorganisms of concern which cause disease can be transmitted in a myriad of ways, whether this occurs as a result of inadequate disinfection of ophthalmic equipment, by contact with contaminated environmental surfaces, or patient to staff transmission.
The pathogens and their associated disease will be explained in this paper along with the importance of using high-level disinfectants including chlorine dioxide in infection prevention.
Common Infections in Ophthalmology and Optometry
Keratitis
This is a condition in which the eye’s cornea becomes inflamed. Symptoms can include moderate to intense pain, impaired eyesight, corneal ulceration, sensitivity to light (photophobia) and red eye.
The causative organisms include bacteria (commonly Pseudomonas aeruginosa and Staphylococcus aureus), parasites (e.g. Acanthamoeba spp.), and fungi (yeasts, moulds and microsporidia)1.
Acanthamoeba keratitis (AK) is a disease of particular concern within ophthalmology. The main route of infection is through the eye, although entry to the body can also occur through the nasal passage and ulcerated skin².
AK is caused by a parasite which feeds off the cornea of the eye.
The parasites which cause AK are ubiquitous within the environment. They are commonly found in contact lens accessories, tap water, dust, and swimming pools and are known to act as hosts for several bacterial species causing infections such as bacterial keratitis and legionellosis. Acting as a host provides protection to pathogens in the aquatic environment against standard water treatment methods.
Symptoms of AK can often be confused with common eye infections such as conjunctivitis and herpes simplex virus keratitis. If infection is left untreated, AK has the ability to cause permanent corneal scarring, and in extreme cases, blindness³.
For medication to have the best chance at effectively treating AK, an early diagnosis for the patient is essential. Individuals who wear contact lenses are most susceptible to contracting AK, although infection can occur in anyone.
Studies have revealed that within the United States approximately 85% of AK infections occur in contact lens wearers. The incidence of the disease in developed countries is approximately one to 33 cases per million contact lens wearers².
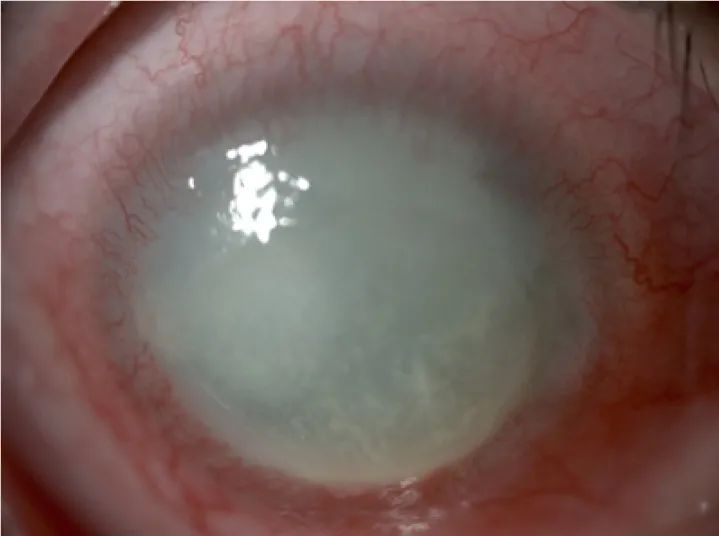
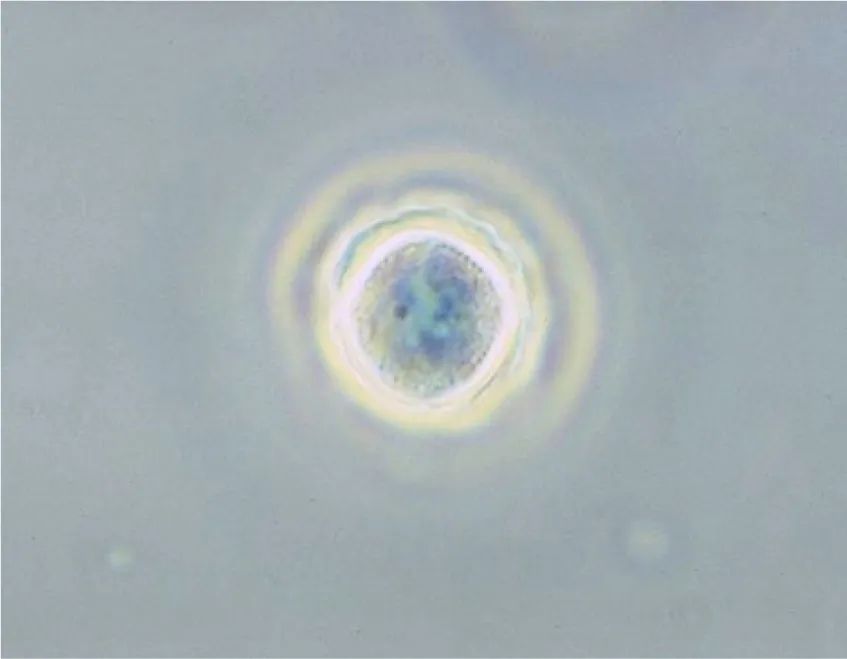
|
Acanthamoeba keratitis. |
Acanthamoeba polyphaga cyst. |
Adenoviral epidemic keratoconjunctivitis
Approximately 92% of acute epibulbar infections are caused by the adenovirus. Adenoviral epidemic keratoconjunctivitis (EKC) is a highly contagious, severe form of conjunctivitis, mostly prevalent in late winter, spring and early summer.
Within larger hospitals (>500 beds), for every 1000 treatment cases, 4.7 are estimated to be from EKC. Major risk factors relating to nosocomial outbreaks of EKC or epidemics include inadequately disinfected ocular instrumentation and insufficient hand washing between healthcare staff and patients⁴.
Human adenovirus (HAdV) is a hostile pathogen as it survives for long periods outside of the human body. It is resistant to adverse pH conditions in the environment and remains stable against many chemical agents.
With studies revealing that serotype HAdV 19 is viable on tonometer tips for nine days and up to 35 on plastics, the necessity for adequate infection control is therefore emphasised5.
Source: https://en.wikipedia.org/wiki/Adenoviral_keratoconjunctivitis
Endophthalmitis
Endophthalmitis is an inflammatory condition of the intraocular cavities, caused by either bacterial or fungal infection. Candida spp. are a common cause e.g. Candida albicans (also implicated with opportunistic oral and genital infections) is the most common cause of endophthalmitis.
Exogenous endophthalmitis arises through the direct introduction of microorganisms (predominantly Candida spp.) into the eye through trauma (including surgery complications) or preceding keratitis. The epidemiological characteristics of patients who contract this infection include:
- Postoperative infection incurred after lens removal
- Postoperative infection incurred after lens implantation
- Postoperative infection incurred after corneal transplant
Endogenous (bloodborne/internal cause) endophthalmitis is the less common form of the disease. This infection occurs through metastatic spread from a distant bodily site⁶.
Other notable infections caused by pathogens of concern in ophthalmology include sty, uveitis, cellulitis, and ocular herpes. Eliminating the initial risk of infection is essential in helping prevent the spread of these pathogens.
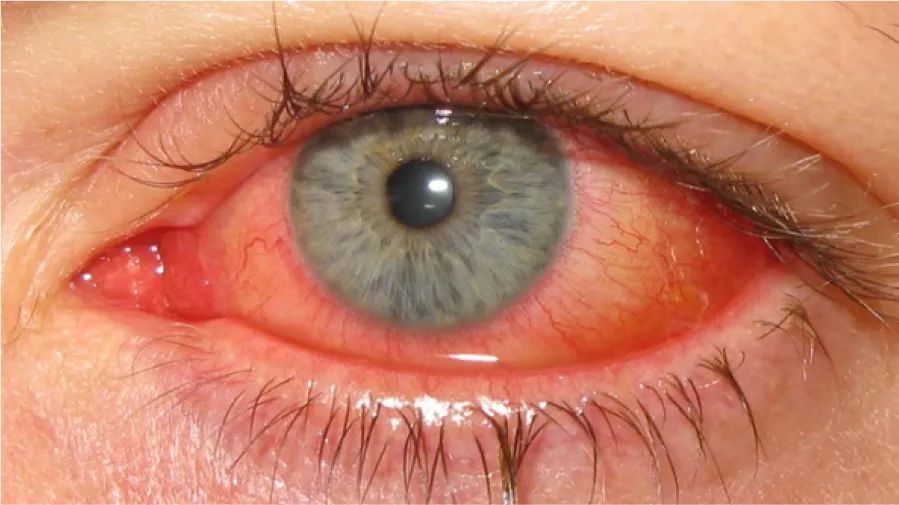
Slit lamp view of endophthalmitis causing hypopyon (inflammation).
Source: https://commons.wikimedia.org/wiki/File:Hypopyon_1.jpg
Tristel’s Role In Ophthalmology and Optometry
Tristel Solutions Ltd produces a range of high-level disinfectants for both medical devices and medical surfaces to reduce the risk of infection.
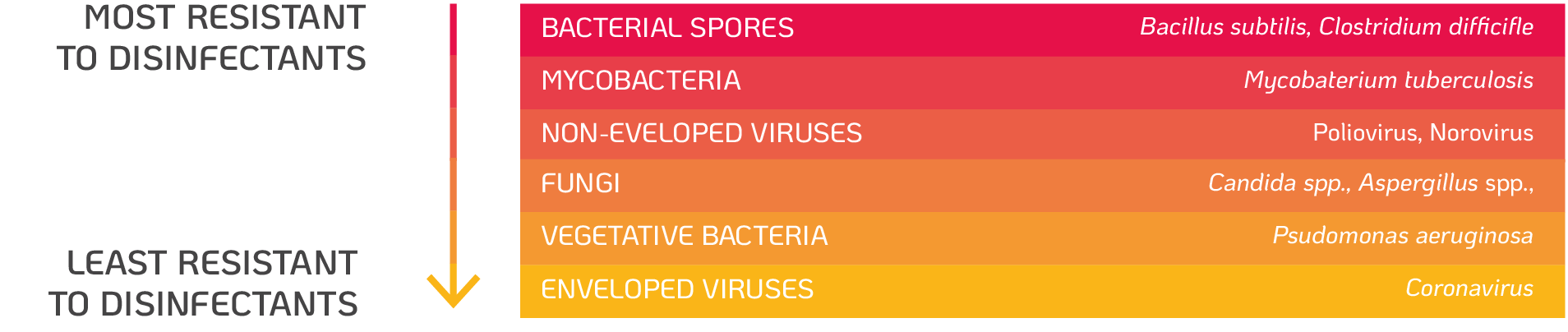
Microorganisms vary in their resistance to disinfectants depending on their molecular structure and composition. Bacterial spores have been shown to be the most resistant, followed by parasite cysts (coccidia), mycobacteria (e.g. M. tuberculosis), non-enveloped viruses (e.g. Adenovirus), fungi (e.g. Aspergillus and Candida spp.), vegetative bacteria (e.g. Pseudomonas spp. and Staphylococcus spp.) and enveloped viruses (e.g. Herpes simplex virus (HSV), coronaviruses) (figure 1.).
Resistance of microorganisms to disinfectants. Adapted from Centers for Disease Control and Prevention (2008)7.
High-level disinfectants (HLDs) kill bacteria, fungi, viruses, mycobacteria and small numbers of the highly resistant bacterial spores.
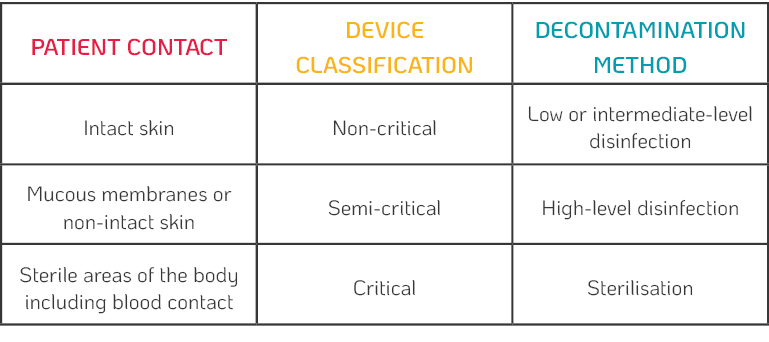
Guidelines referring to the disinfection of reusable ophthalmic devices are usually written by professional advisory bodies or government departments. Differences can be found from one country to another, however, devices coming in direct contact with the eye are always classified as semi-critical.
According to the Spaulding classification (Table 1.), semi-critical devices require high-level disinfection because they touch mucous membranes or non-intact skin (which is true of 99% of ophthalmic devices).
Tristel’s proprietary chlorine dioxide formula is a broad-spectrum biocide with proven efficacy against a wide range of microorganisms such as bacteria, viruses, parasites (protozoa cysts), fungi, mycobacteria and bacterial spores.
Chlorine dioxide is a potent oxidative biocide. Oxidation is the removal of electrons from a microorganism leading to its death. The advantage of this mode of action over non-oxidising biocides such as alcohols, aldehydes, and quaternary ammonium compounds, is the inability for microorganisms to develop resistance against chlorine dioxide, as well as its broad range of efficacy.
This unique formulation has been developed to balance concentration for rapid efficacy, and excellent material compatibility with devices.
Tristel’s Chlorine Dioxide Has Proven Efficacy
Products using Tristel’s chlorine dioxide formulation have been tested in accordance with European standard EN 14885:2018 which provides a framework for testing the microbiocidal activity of chemical disinfectants. The products tested in accordance to this standard are intended for use in human medicine, which includes ophthalmology and optometry.
The microorganisms chosen by scientists for testing under each vertical standard within EN 14885:2018, are representative of the most resistant microorganisms to disinfectants in the intended area of use. Efficacy against these mandatory organisms infers efficacy against all non-tested microorganisms, including emerging pathogens. For example, efficacy against mandatory viruses Poliovirus Type 1, Adenovirus Type 5 and Murine Norovirus in accordance with EN 14476 infers virucidal activity against all other enveloped and non-enveloped viruses including SARS-CoV-2 (the virus responsible for the COVID-19 pandemic). Tristel’s chlorine dioxide has now been proven to destroy the real life pathogenic SARS-CoV-2 in 30 seconds in the presence of soiling*.
Currently no standard test norms have been developed for testing against the problematic Acanthamoeba spp., therefore Tristel has carried out bespoke testing against Acanthamoeba castellanii cysts to ensure specific efficacy of our proprietary chlorine dioxide formulation.
A. castellanii is one of the strains which cause AK.
The cyst form of A. castellanii resembles the highly resistant spore form of bacteria, therefore this bespoke testing was performed according to a sporicidal test method widely used in European testing standards (EN 13704).
Two chlorine dioxide based HLDs in the ophthalmological sector are Tristel Duo OPH for ophthalmic devices and JET for medical surfaces.
Both products are proven sporicidal, mycobactericidal, virucidal, fungicidal, bactericidal, effective against Acanthamoeba cysts and have been tested in accredited laboratories worldwide.
*Tested in accordance with EN 14476:2013+A2:2019, at a concentration of 20 parts per million (ppm) by a Biosafety category 3 laboratory. All products have a concentration of chlorine dioxide greater than 20ppm at the point of use.
 |
… |
Tristel Duo OPHCategorised as a Class IIa Medical Device, Tristel Duo OPH is designed specifically for the high-level disinfection of ophthalmic medical devices in 30 seconds, including: • Diagnostic contact lenses |
The disinfection of tonometer prisms and contact glasses using Tristel Duo OPH is recommended by Haag Streit. Globally, Haag Streit is one of the largest manufacturers of ophthalmologic equipment.
Ophthalmologic equipment used within the healthcare industry has the potential to harbour and spread a variety of infections. This is particularly hazardous with microorganisms such as Acanthamoeba, due to their ability to act as hosts to other microorganism species. Acinetobacter, Enterobacter, Legionella, various mycobacteria, Pseudomonas and Serratia spp. are examples of microorganisms able to resist disinfectant methods and replicate after ingestion by Acanthamoeba spp.⁸.
Therefore, as Tristel Duo OPH has proven efficacy against Acanthamoeba spp. ophthalmologists can be assured of reliable and quick disinfection.
|
|
… |
JETJET is a powerful sporicidal disinfectant, suitable for use on high-touch medical surfaces in a universal contact time of one minute. JET is also a powerful cleaner destroying RNA and DNA in seconds. JET is a rapid and reliable safeguard for patients and ophthalmology staff wherever the risks of infection are highest. |
COVID-19 Guidelines Recommend Chlorine Dioxide for Infection Control
Regulatory bodies have recognised the potential risk of nosocomial transmission of SARS-CoV-2 via ophthalmic devices and surfaces in this area. In Australasia two regulatory bodies have been proactive in issuing guidelines to help those working in the healthcare environment.
Firstly, The Royal Australian and New Zealand College of Ophthalmologists (otherwise referred to as RANZCO) state that for slit lamps, tonometers, contact lenses, pinhole occluders or any other equipment that has come into close contact with the patient or mucosal surfaces, should be cleaned with a chlorine dioxide disinfectant immediately after seeing a suspect or confirmed case⁹.
Secondly, Optometry Australia wrote that in order to adhere to the recommendation that semi-critical devices (tonometer probes) must obtain high-level disinfection, the gold standard for disinfection would involve either Tristel Duo OPH or sodium hypochlorite10.
Conclusion
The infections and microorganisms causing disease described in this paper can be difficult to diagnose and differentiate from one another. They can also lead to serious complications.
Infection prevention by the Ophthalmologist should include:
• Proper hand washing between each consultation, especially if the patient’s cornea has been touched
• High-level disinfection of the devices before starting another procedure
The active ingredient of many Tristel products is a proprietary chlorine dioxide formulation. These products are situated in the Tristel Portfolio for medical device disinfection, including the Tristel Trio Wipes System and Tristel Duo, as well as The Cache Collection comprising environmentally conscious surface cleaners and disinfectants, including JET and FUSE.
References
1. S. Tuft MChir and M. Burton, “Focus – Microbial keratitis,” London, 2013
2. Centers for Disease Control and Prevention, “Parasites – Acanthamoeba – Granulomatous Amebic Encephalitis (GAE); Keratitis,” 2017. https://www.cdc.gov/parasites/acanthamoeba/index.html (accessed Sep. 01, 2020).
3. All About Vision and C. A. Knobble, “Corneal Ulcer: Causes And Treatment,” 2016. https://www.allaboutvision.com/conditions/corneal-ulcer.htm (accessed Sep. 01, 2020).
4. A. Bialasiewicz, “Adenoviral Keratoconjunctivitis,” Sult. Qaboos Univ. Med. J., 2007, [Online]. Available: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3086413/.
5. Centers for Disease Control and Prevention (CDC), “Adenovirus-associated epidemic keratoconjunctivitis outbreaks–four states, 2008-2010.,” MMWR. Morb. Mortal. Wkly. Rep., vol. 62, no. 32, pp. 637–51, 2013, [Online]. Available: http://www.ncbi.nlm.nih.gov/pubmed/23945769%0Ahttp://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=PMC4604776.
6. S. A. Klotz, C. C. Penn, G. J. Negvesky, and S. I. Butrus, “Fungal and Parasitic Infections of the Liver,” Clin. Microbiol. Rev., vol. 13, no. 4, pp. 662–685, 2000.
7. Centers for Disease Control and Prevention, “Guideline for Disinfection and Sterilization in Healthcare Facilities, 2008; Miscellaneous Inactivating Agents,” CDC website, no. May, pp. 9–13, 2008, doi: 1.
8. V. Thomas, G. McDonnell, S. P. Denyer, and J. Y. Maillard, “Free-living amoebae and their intracellular pathogenic microorganisms: Risks for water quality,” FEMS Microbiol. Rev., vol. 34, no. 3, pp. 231–259, 2010, doi: 10.1111/j.1574-6976.2009.00190.x.
9. RANZCO, “RANZCO-Coronavirus-COVID-19-Guideline,” 2020.
10. Optometry Australia, “Infection Control and COVID-19 factsheet,” 2020. [Online]. Available: http://www.waht.nhs.uk/en-GB/Our-Services1/Departments/Infection-Control/Infection-Control-and-MRSA/.
©2021 Tristel Solution Limited. All rights reserved.