Triple-enzyme foam for Medical Device Cleaning
Designed specifically for use on non-lumened invasive and non-invasive medical devices.
Product Code: TSL023301

Applications
Tristel Clean can be used as a standalone cleaning product, or prior to disinfection with Tristel Duo OPH.
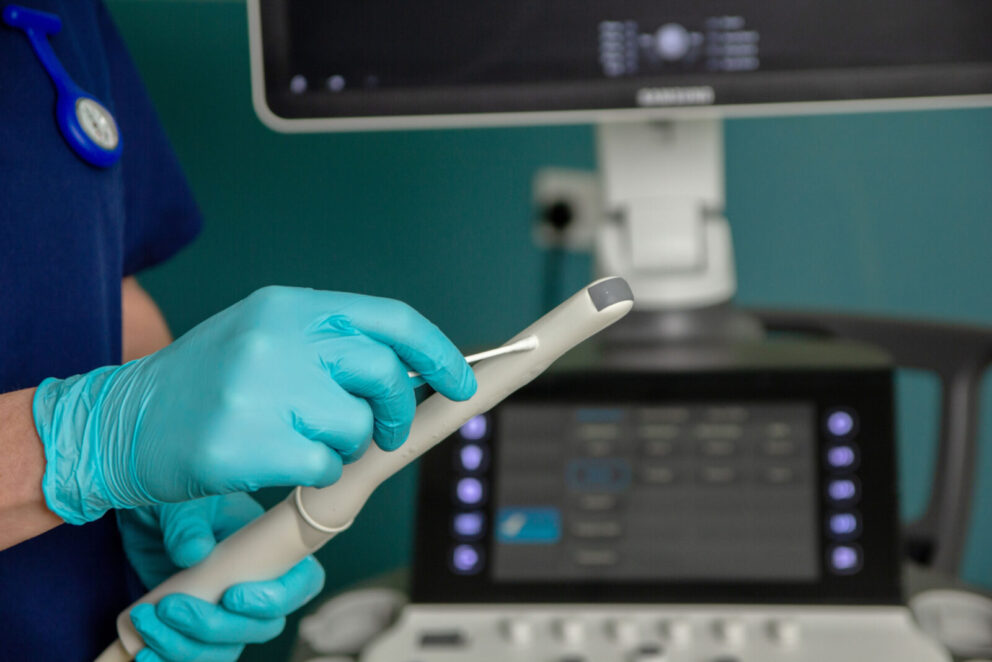
Endocavity Probes
Skin Surface Transducers
Manometry Catheters
Ophthalmic Devices
Laryngoscopes
Why Choose Tristel Clean?
Disinfection is only effective when the probe has undergone thorough cleaning.
- Long Lasting
- Compatible
- Mobile and Practical
-
Tristel Clean has a 2-year shelf life, ensuring that hospitals and clinics can maintain compliance with infection prevention protocols without interruption, even in low-usage areas.
-
Tristel’s cleaning chemistry has been approved by most major manufacturers for use on their devices, including:
- Advanced Tactile Imaging
- Arthrex
- BK Medical
- Canon Medical Systems
- Fujifilm Healthcare
- GE Healthcare
- Philips
-
Lightweight, easy-to-use and designed for point-of-care applications. Requires no soaking, making Tristel Clean convenient for healthcare settings.
Additionally, Tristel Clean features a non-aerosol application that produces foam, ensuring the cleaning agent adheres effectively to medical equipment. This delivers efficient cleaning and allows precise control over the amount of product applied.
Product information
- Description
- Easy to Use
-
Description
Each 240ml Tristel Clean bottle contains 310 doses of foam and is supplied in cases of 6 bottles.
When using Tristel Clean, no intermediate rinsing is required when using Tristel Duo OPH for high-level disinfection.
Tristel Clean is classified as Class I Medical Device under the Canadian Medical Device Regulations SOR/98-282.
-
Easy to Use
Refer to the User Guide for full instructions.
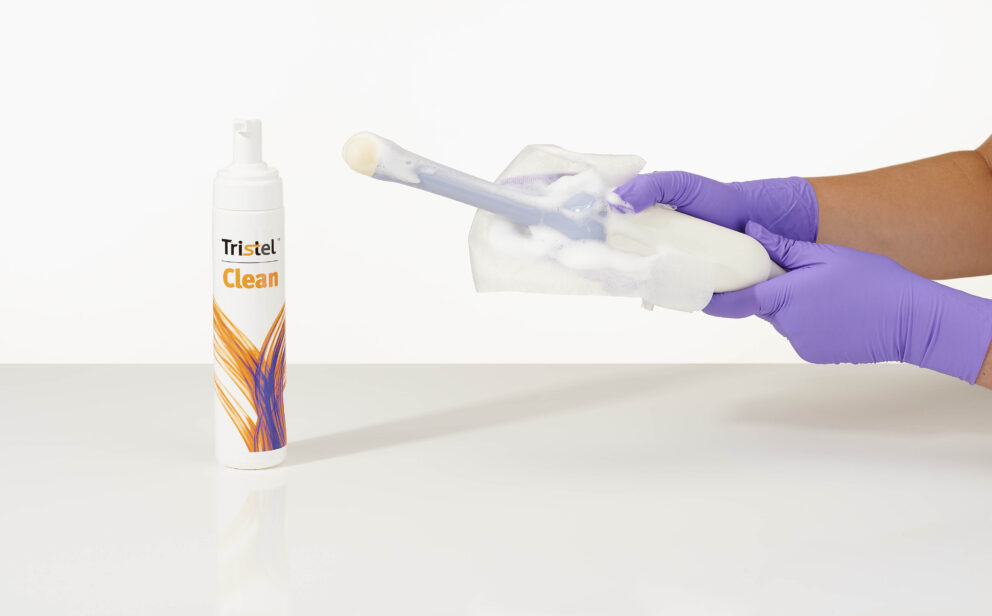
STEP 1: Dispense
Dispense Tristel CLEAN onto a dry wipe. DUO WIPES are recommended.
STEP 2: Cleaning
Wipe the medical device to remove all visible soil.